Table of Contents
What is non-muscle-invasive bladder cancer?
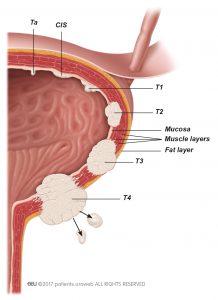
Non–muscle-invasive bladder cancer, also called superficial bladder cancer, is a superficial cancer that has not grown into deeper layers of the bladder wall (Fig. 1).
There are three subtypes based on how they grow:
- Stage Ta tumours are confined to the bladder lining.
- Stage T1 tumours have invaded the connective tissue under the bladder lining but have not grown into the muscle of the bladder wall.
- Stage Tis tumours are flat velvet-like tumours, also known as carcinoma in situ (CIS), that are confined to the bladder wall but have potentially a high risk of spreading into the deeper layers of the bladder.
Stage Ta and T1 tumours usually appear as small grapelike growths (also called papillary) that grow toward the centre of the bladder without growing into the deeper bladder layers. Your surgeon may remove these growths using a method called transurethral resection of bladder tumour (TURBT).
Treatment options for non-muscle invasive bladder cancer
Transurethral resection of bladder tumour
TURBT is the surgical removal (resection) of bladder tumours. This procedure is both diagnostic and therapeutic. It is diagnostic because the surgeon removes the tumour and all additional tissue necessary for examination under a microscope (histological assessment). TURBT is also therapeutic because complete removal of all visible tumours is the treatment for this cancer. Complete and correct TURBT is essential for good prognosis. In some cases, a second surgery is required after several weeks.
Instillation treatment
As noted earlier, TURBT can eradicate stage Ta or T1 tumours, but some tumours commonly recur and can progress to invasive cancer. Intravesical (within the bladder) instillation treatment after TURBT should be considered for all patients.

